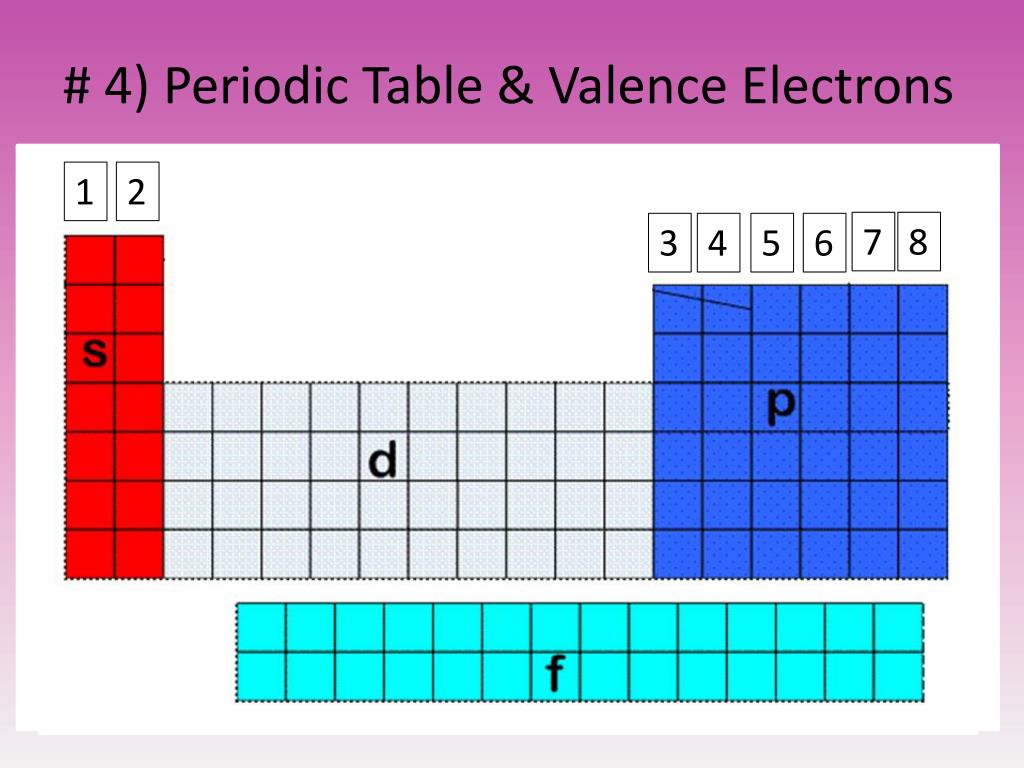
They all have a similar electron configuration in their valence shells: a single s electron. Their electron configurations (abbreviated for the larger atoms) are as follows, with the valence shell electron configuration highlighted: Electrons, electron configurations, and the valence shell electron configuration highlighted. For example, take the elements in the first column of the periodic table: H, Li, Na, K, Rb, and Cs. If we look at just the valence shell’s electron configuration, we find that in each column, the valence shell’s electron configuration is the same. (The inner electrons are called core electrons.) The valence electrons largely control the chemistry of an atom. The electrons in the highest-numbered shell, plus any electrons in the last unfilled subshell, are called valence electrons the highest-numbered shell is called the valence shell. These dots are arranged to the right and left and above and below the. The number of dots equals the number of valence electrons in the atom. The periodic table is separated into blocks depending on which subshell is being filled for the atoms that belong in that section. Valence electrons: The electrons present in the outermost orbit of an atom are called valence electrons. A Lewis electron dot diagram (or electron dot diagram, or a Lewis diagram, or a Lewis structure) is a representation of the valence electrons of an atom that uses dots around the symbol of the element. Explain the long form of the periodic table.\): Blocks on the Periodic Table.This electron arrangement indicates that the outermost orbit of Aluminum element (Al) has 3 electrons. Hence the aluminum element has electrons arrangement 2, 8, 3. How does the number of valence electrons vary on moving from left to right:(i) In the first period of the periodic table? (ii) In the second period of the periodic table? Now the atomic number of aluminum (Al) is 13.Consider the following elements:Na, Ca, Al, K, Mg, Li (a) Which of these elements belong to the same period of the periodic table? (b) Which of these elements belong to the same group of the periodic table?.5.17: Electron Configurations and the Periodic Table. (a) What is the main characteristic of the last elements in the periods of the periodic table? What is the general name of such elements?(b) What is the number of elements in: (a) 1st period, and (b) 3rd period, of the modern periodic table? ChemPRIME (Moore et al.) 5: The Electronic Structure of Atoms.Define the Periodic Table with elements name. Figure 1: Periodic Table (Free Printable periodic tables (pdf and PNG) 2023) Explain the significance of the S, P and D blocks in the periodic table.

/PeriodicTableCharge-BW-56a12db13df78cf772682c34.png)
(a) What is the usual number of valence electrons and valency of group 18 elements of the periodic table?(b) What happens to the number of valence electrons in the atoms of elements as we go down in a group of the periodic table?.(a) How does the electropositive character of elements change on going down in a group of the periodic table?(b) State how the valency of elements varies (i) in a group, and (ii) in a period, of the periodic table. Try our Interactive Periodic Table of Elements that lets you sort by metals, physical states, group, and period, and view the most important elemental properties.In the modern periodic table, which are the metals among the first ten elements?.Compare and contrast the arrangement of elements in Mendeleev’s Periodic Table and the Modern Periodic Table.The elements in a group have very similar chemical properties, which arise from the number of valence electrons presentthat is, the number of electrons in the outermost shell of an atom. What is the major characteristic of the first elements in the periods of the periodic table? What is the general name of such elements? Periodic table, in chemistry, the organized array of all the chemical elements in order of increasing atomic number.Name of 118 elements in the periodic table.How does the valency of elements change on moving from left to right in the third period of the periodic table?.How does the valency of elements vary in going down a group of the periodic table?.
